BYCROSS
Atherectomy & Thrombectomy Catheter

Venous Stent
The blueflow Venous Stent is indicated for the treatment of venous outflow obstructions in the lower extremities. With its closed cell design, blueflow Venous Stent is highly flexible.The blueflow Venous Stent is available in different configurations.

Many people suffer from venous diseases. Varicose veins and spider veins are terms that almost everyone knows. If varicose veins or even venous ulcers (Ulcus Cruris) occur again despite a varicose vein treatment, these are often the consequences of an obstruction in the pelvic vein caused by a thrombosis.
In the course of thrombosis in the pelvic vein, scarring and obstruction often occur. Since the venous valves are involved, they can lose their function. This leads to blood congestion in the legs.
Typical consequences of pelvic vein obstruction or occlusion are varicose veins, painful swelling of the legs, calf cramps and poorly healing venous ulcers.
Modern, non-invasive diagnostic procedures can precisely locate these obstructionand occlusions.
If an acute or chronically occluded or severely obstructed pelvic vein has been diagnosed as the cause, it can be reopened by a minimally invasive catheter procedure.
In the pelvic vein area, a venous stent is then advanced under radiological control up to the narrow point and released there. In the case of extensive occlusions, it is sometimes necessary to implant several or one long stent.
Due to the reopening of the pelvic vein the blood flow in the direction of the heart is improved. This reduces the backflow in the legs and thus the pressure on the tissue. After this treatment, open ulcers have the chance to heal and massive swelling of the legs can be reduced.The reopening of occluded pelvic veins is a complex procedure that requires a great deal of experience and a venous stent that fulfils special requirements.
These minimally invasive procedures are performed in special centers.
The blueflow Venous Stent was developed and produced in Germany in cooperation with leading international vein specialists. It is now marketed worldwide.
The blueflow Venous Stent is characterized by its extreme flexibility and kink and compression resistance. Due to the special processing technology, the blueflow Venous Stent adapts to the natural course of the vessel and does not kink during everyday movements. Maximum compression resistance and flexibility are the most important criteria for venous stents implanted below the inguinal ligament.
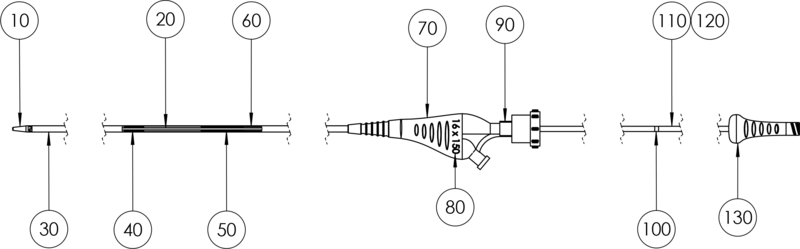
10 Tip, radiopaque | 20 Guide wire lumen | 30 Shaft, stainless steel enforced & braided | 40 Retriever |
50 Stent Pusher (with Pt / Ir- Marker band) | 60 Pusher Tube | 70 Handle, distal | 80 Size Printing | 90 Tuohy-Borst Valve |
100 Marker Retriever | 110/120 Metal Pusher Tubes | 130 Handle, proximal
1. blueflow Venous Stent
The blueflow Venous Stent is a self-expanding stent. It is made of woven Nitinol (nickel titanium alloy) wires in a closed-loop design.
2. blueflow Venous Stent Delivery System
The stent is preloaded in a 10 Fr delivery system with a shaft lengsth of 100 cm.
3. Recommended materials (not included in the set)
• Shaft length 100 cm
• Recommended Guidewire 0.035˝
• Recommended introducer sheath 10 Fr
blueflow is a trademark of plus medica GmbH & Co. KG
Contact us
Click here, to find out more about the publication.
Click here to read the publication.
| Art. No. | Description | VE |
|---|---|---|
| VS12060 | blueflow Venous Stent 12 × 60 mm | 1 |
| VS12100 | blueflow Venous Stent 12 × 100 mm | 1 |
| VS14060 | blueflow Venous Stent 14 × 60 mm | 1 |
| VS14100 | blueflow Venous Stent 14 × 100 mm | 1 |
| VS14150 | blueflow Venous Stent 14 × 150 mm | 1 |
| VS16060 | blueflow Venous Stent 16 × 60 mm | 1 |
| VS16100 | blueflow Venous Stent 16 × 100 mm | 1 |
| VS16150 | blueflow Venous Stent 16 × 150 mm | 1 |
| VS18060 | blueflow Venous Stent 18 × 60 mm | 1 |
| VS18100 | blueflow Venous Stent 18 × 100 mm | 1 |